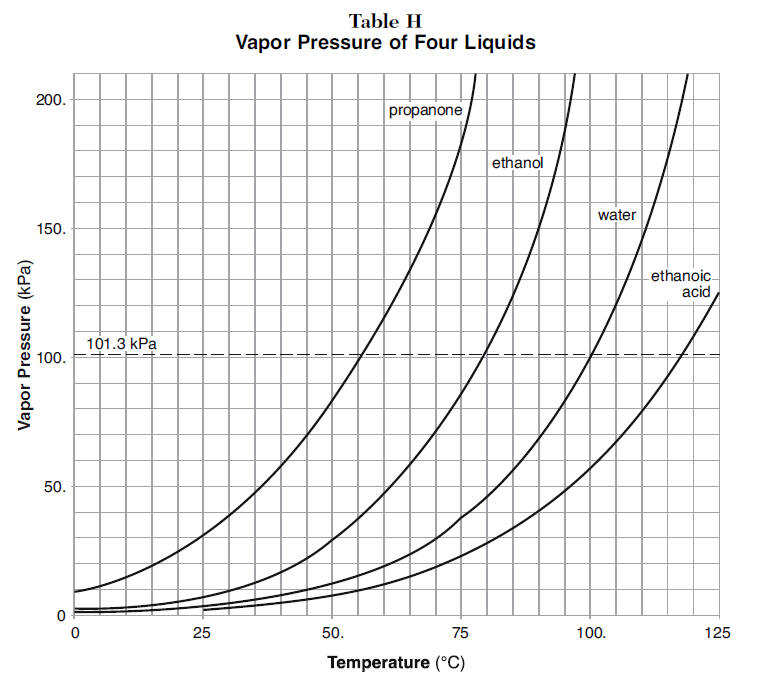
Calculate the boiling point of water at 24 torr pressure. The average Hvap. over the temperature range is 10.12 kcal mol^-1 . Will all the water form gaseous state if placed in
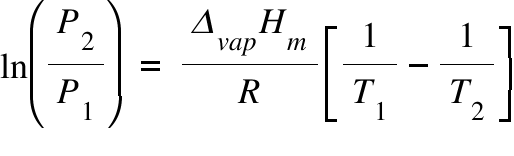
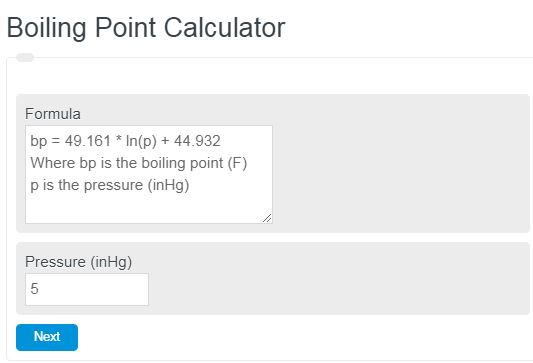
Correlation of Vapor Pressure at Different Temperatures by Clausius Clapeyron Equation Calculator | Calistry

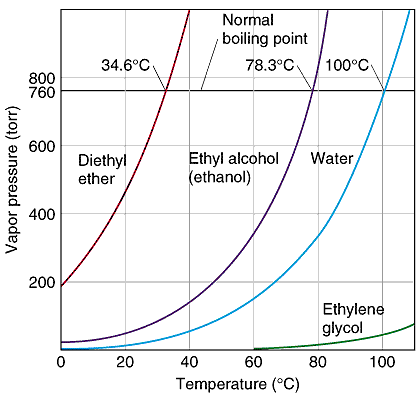
OneClass: Calculate the normal boiling point of a compound that has a vaporpressure of 500 torr at 20...
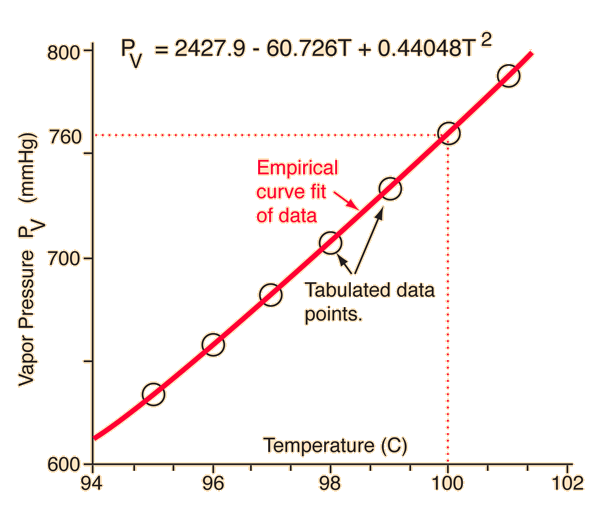
Problem Set #10 Assigned November 8, 2013 – Due Friday, November 15, 2013 Please show all work for credit To Hand in 1.

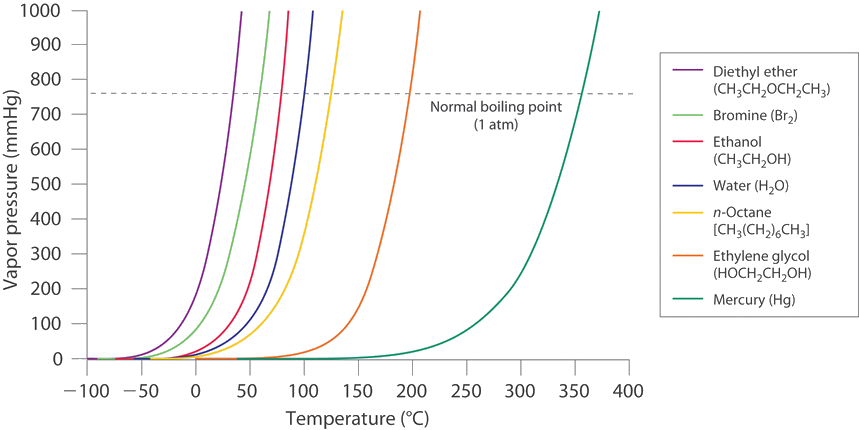
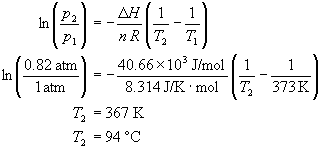
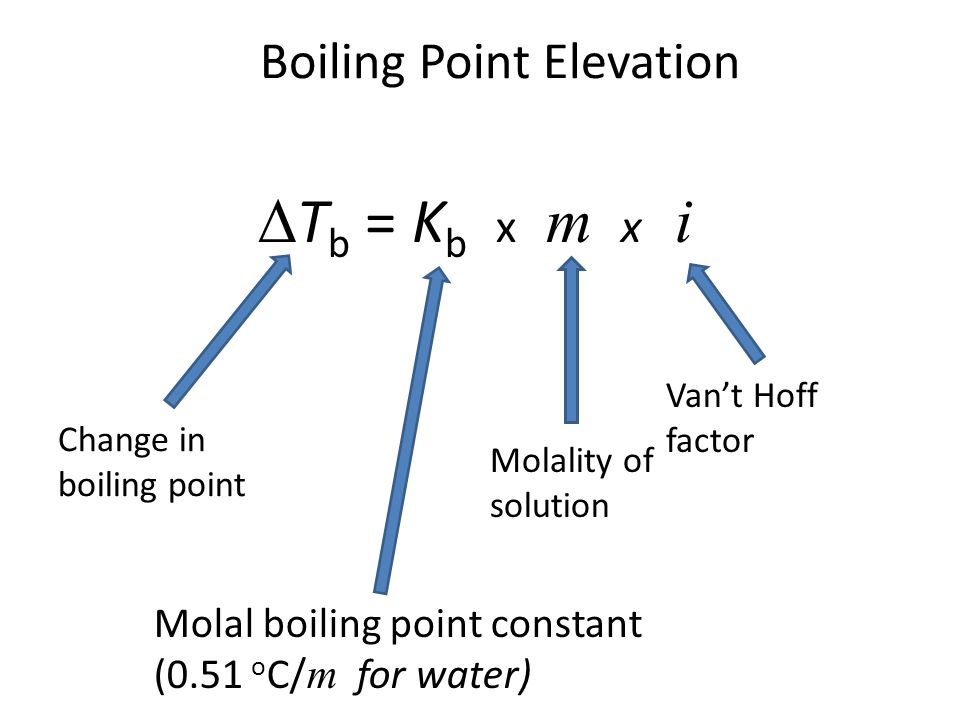
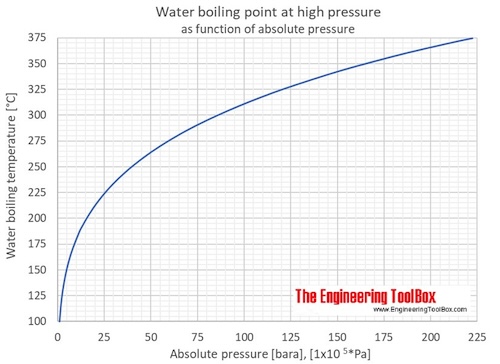


![Nomograph for the calculation of boiling points under vacuum - [www.rhodium.ws] Nomograph for the calculation of boiling points under vacuum - [www.rhodium.ws]](https://www.designer-drug.com/pte/12.162.180.114/dcd/chemistry/equipment/pictures/nomograph.gif)