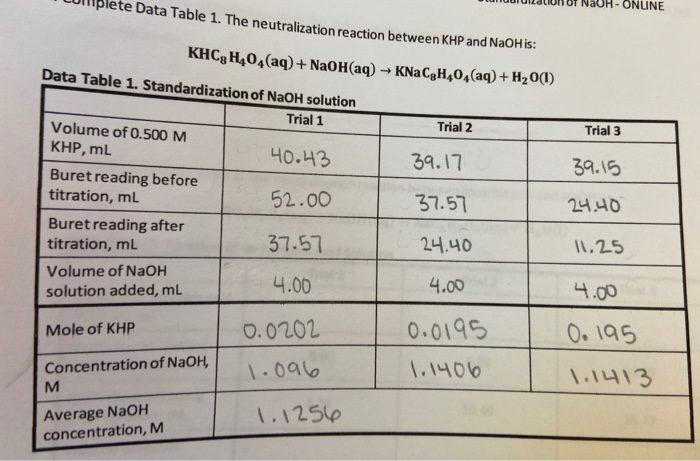
Calculate the molarity of NaOH in the solution prepared by dissolving its 4 g in enough water to form 250 mL of the solution.
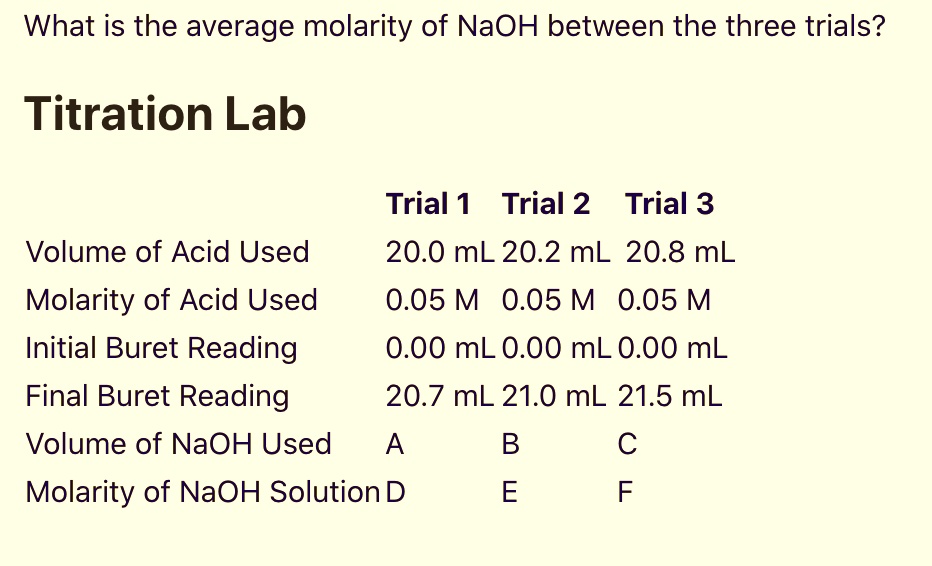
SOLVED: What is the average molarity of NaOH between the three trials? Titration Lab Trial 1 Trial 2 Trial 3 20.0 mL 20.2 mL 20.8 mL 0.05 M 0.05 M 0.05 M

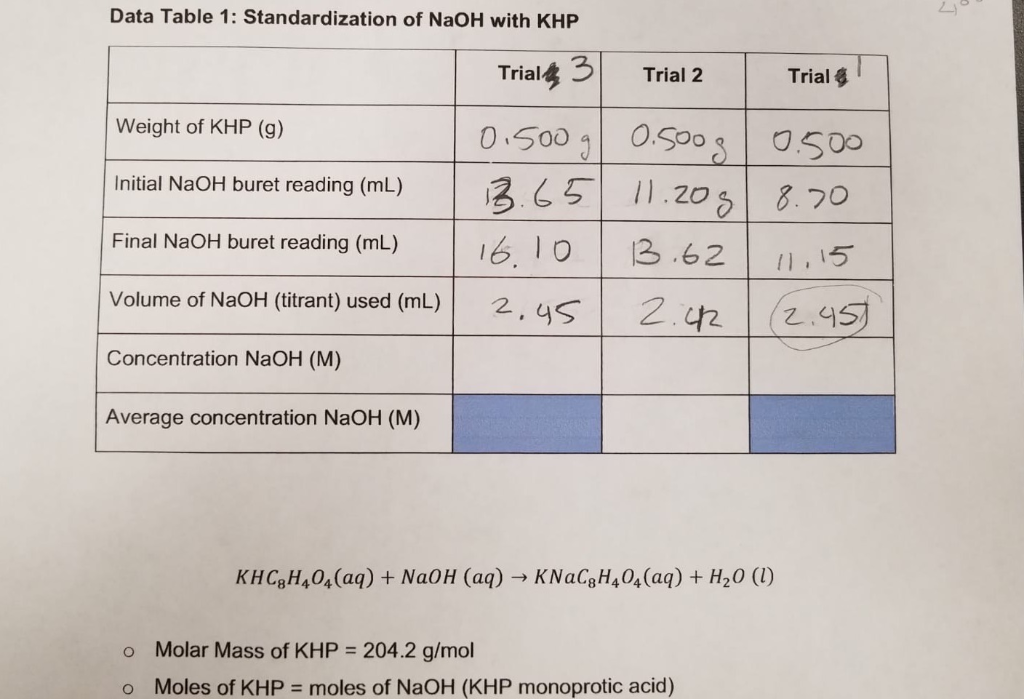
SOLVED: Calculations and questions (15points) Standardization of NaOH Volume of NaOH used 18.3mL 18.6 mL -17.8 mL Trial #1 Trial #2 Trial #3 (if > 3 trials, indicate additional volume(s) On other

Calculate molarity of NaOH in a solution made by mixing 2 L of `1.5 M NaOH,3 L` of 2 M NaOH and 1L - YouTube

How to Calculate Analyte Concentration Using the Equivalence Point in an Acid-base Titration | Chemistry | Study.com

SOLVED:It took 25.06 ±0.05 mL of a sodium hydroxide solution to titrate a 0.4016-g sample of KHP (see Exercise 79). Calculate the concentration and uncertainty in the concentration of the sodium hydroxide





![Calculating pH from [OH-] hydroxide Concentration - CLEAR & SIMPLE - YouTube Calculating pH from [OH-] hydroxide Concentration - CLEAR & SIMPLE - YouTube](https://i.ytimg.com/vi/gn1CgBzShps/maxresdefault.jpg)

:max_bytes(150000):strip_icc()/how-to-calculate-normality-609580final2-0d5efa5a961f4fa0a7efc780921faee1.png)