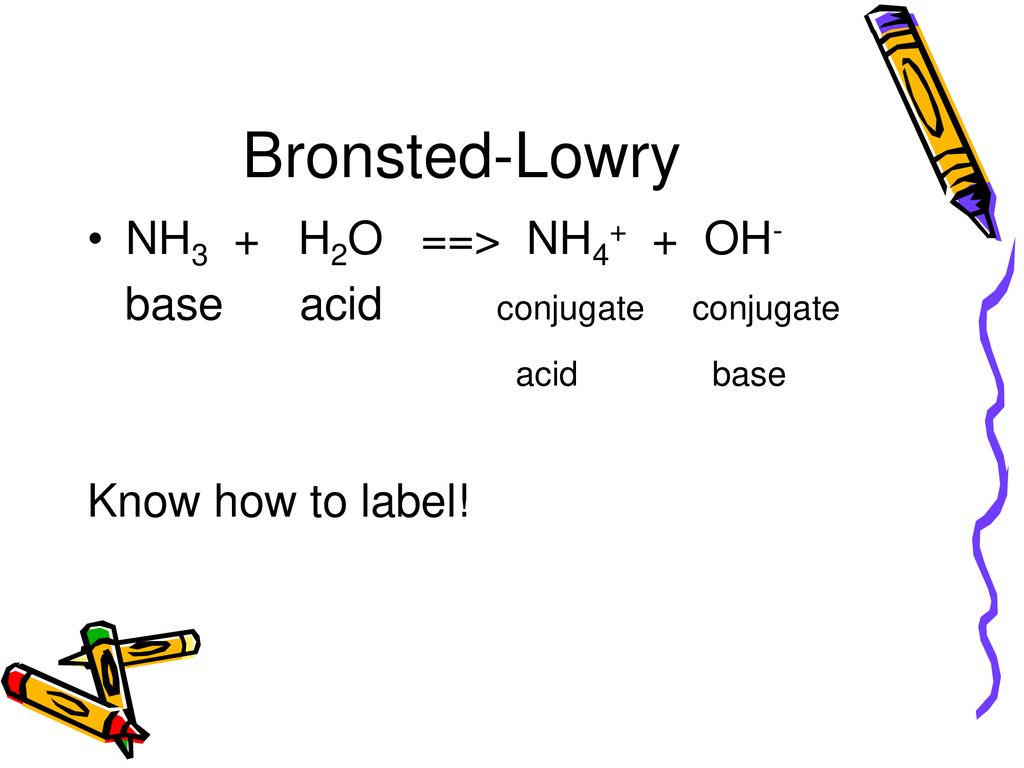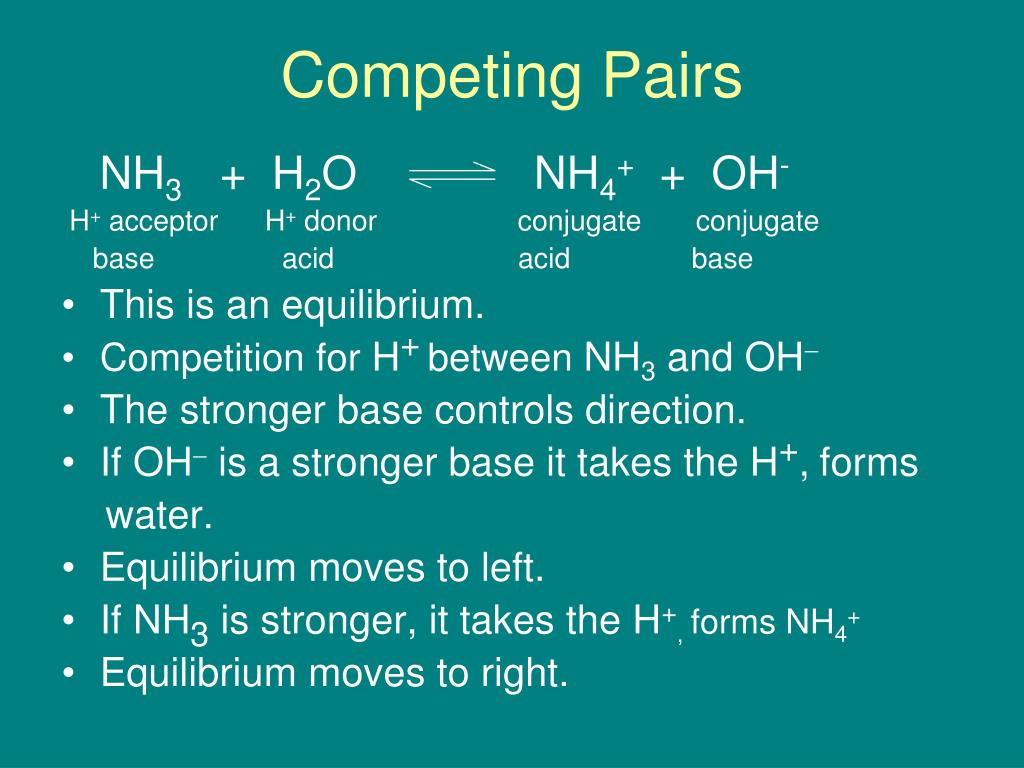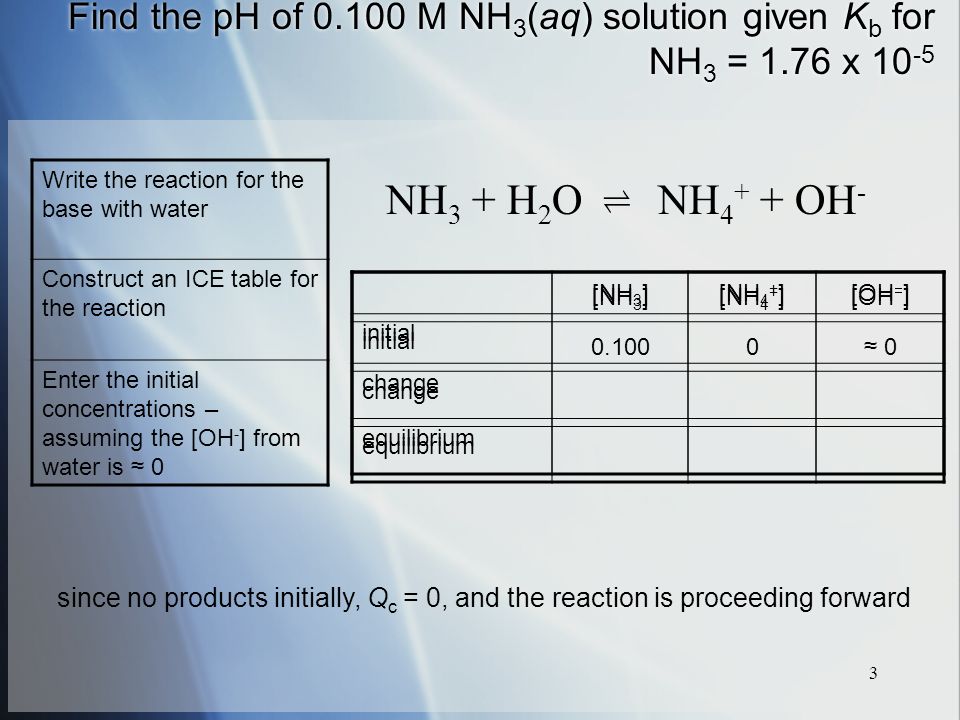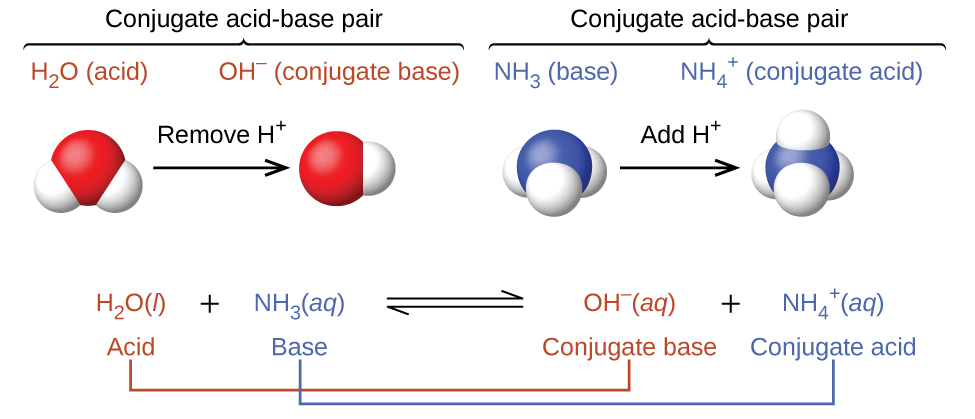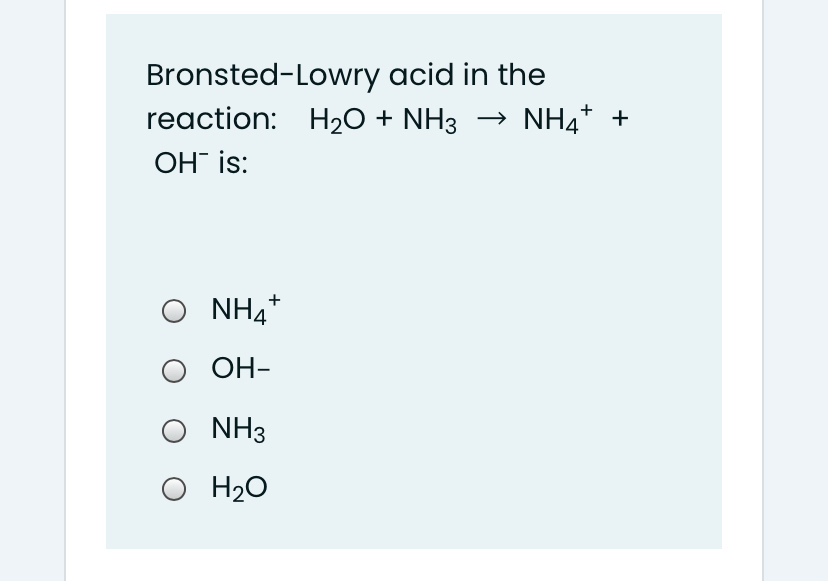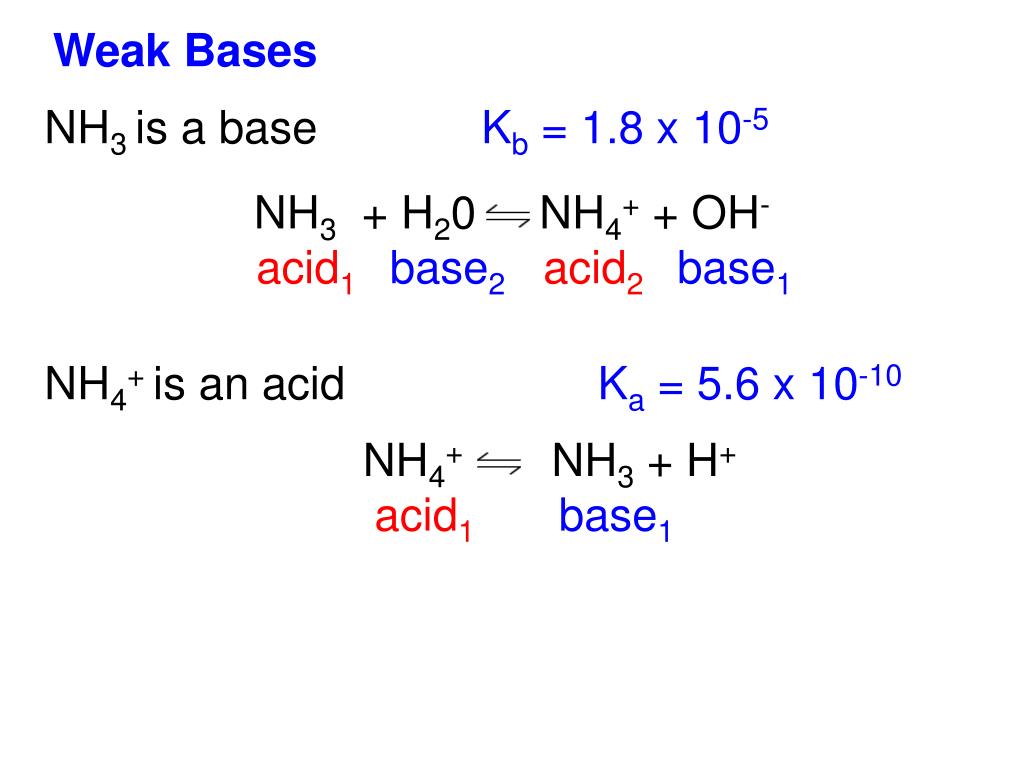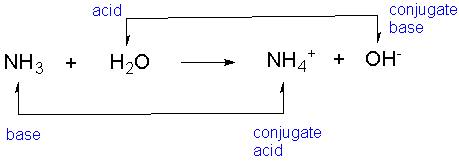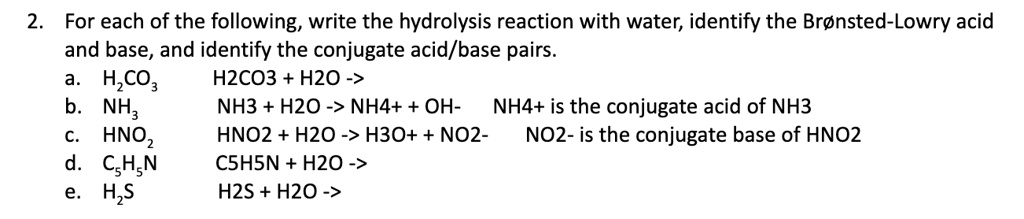
SOLVED: For each of the following, write the hydrolysis reaction with water; identify the Bronsted-Lowry acid and base, and identify the conjugate acid/base pairs HZCO; H2CO3 H2O b. NH; NH3 + H2O ->
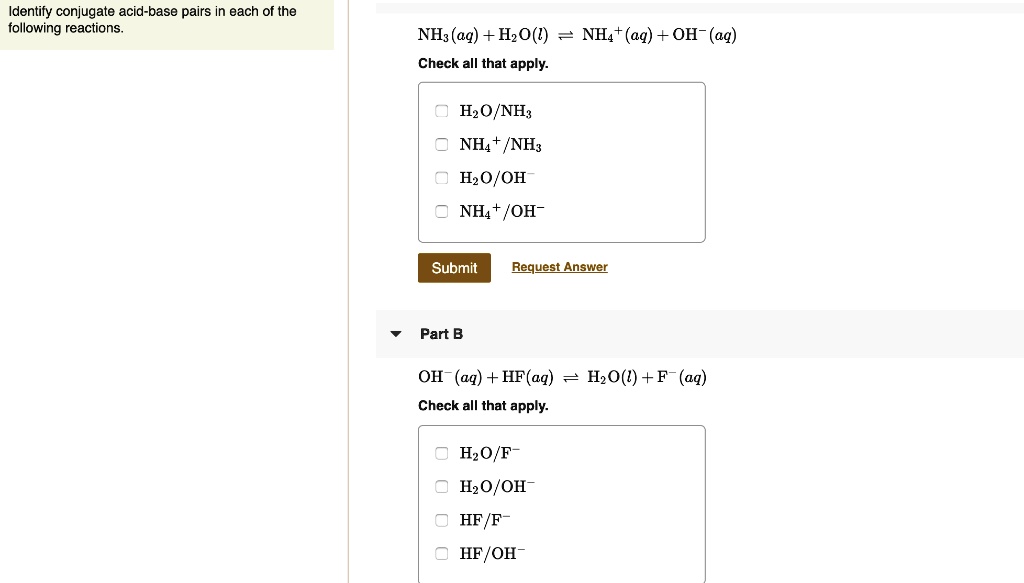
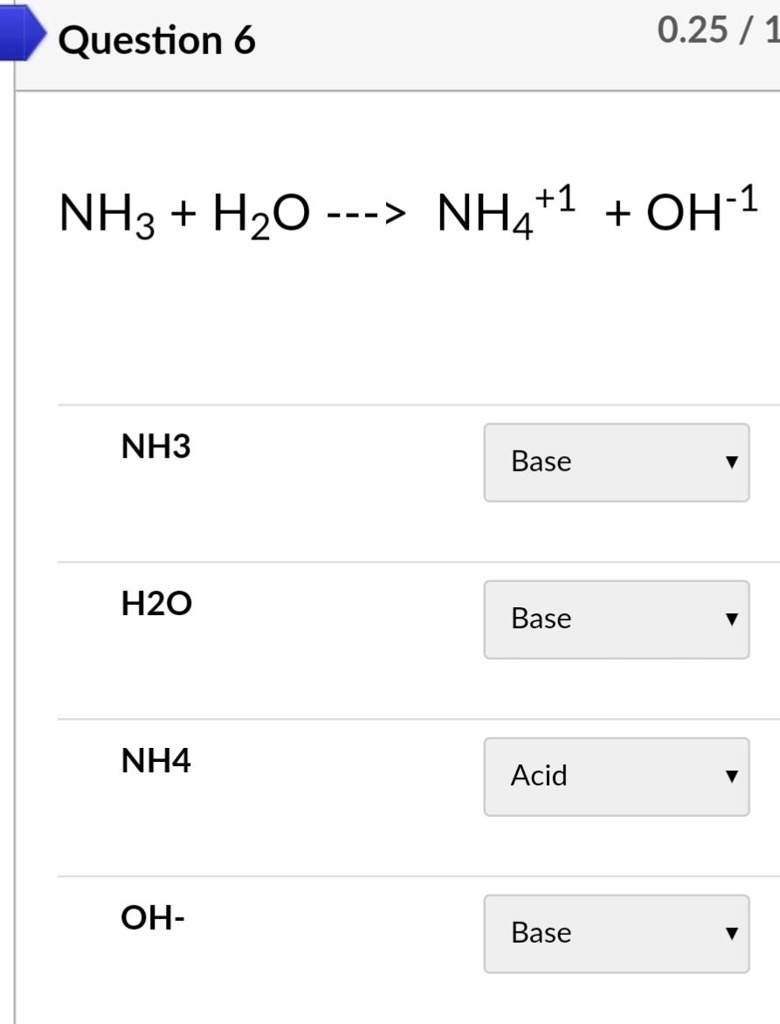
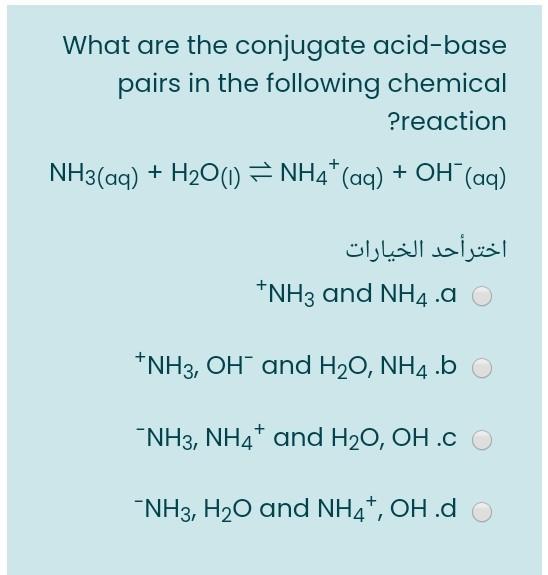
SOLVED: Identify conjugate acid-base pairs in each of the following reactions NH; (aq) + HzO() Check all that apply: NH4 + (aq) + OH- (aq) HzO/NH: NH4 NHz HzO/OH NH4 + /OH-