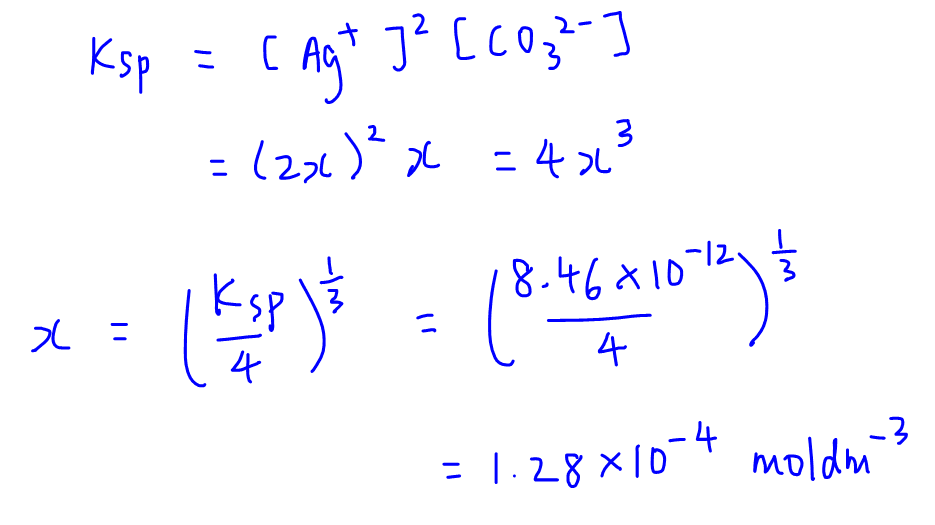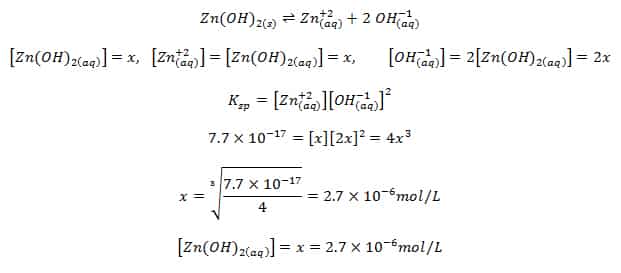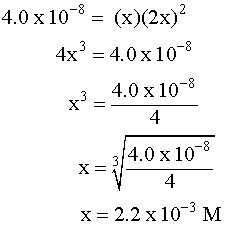SOLVED: Calculate the solubility (in g/L) of O2 in water at a partial pressure of O2 of 120 torr at 25C (K o2 = 780 atm*L/mol)

Calculate solubility (in moles / litre) of a saturated aqueous solution of Ag3PO4 if the vapour pressure of the solution becomes 750 torr at 373 K(Assume molality = molarity).
the solubility of CaCO3 is 7*10^ 3 mg/litre.calculate the solubility of BaCO3 (in mol/l) from this information and from the fact that when Na2CO3 is added slowly to a solution containing equimolar